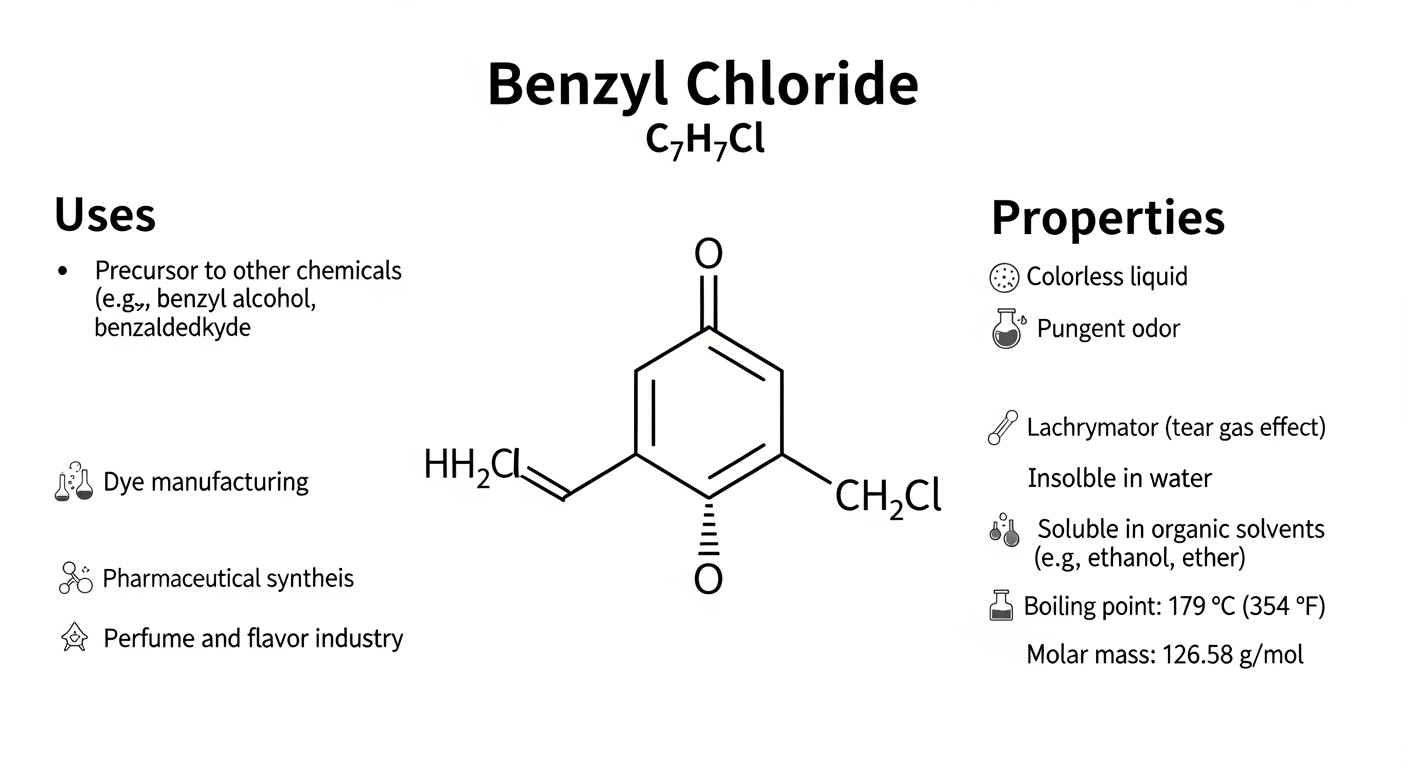
Benzyl chloride is an important organic compound widely used in chemical manufacturing and research. It belongs to the family of aromatic compounds and plays a key role as an intermediate in many industrial processes. Chemists often study benzyl chloride because of its reactive nature and its ability to form various other useful chemicals. The compound is typically used in the production of pharmaceuticals, dyes, and synthetic materials. Understanding benzyl chloride involves exploring its structure, properties, production methods, and applications. By learning about this compound, readers can gain insight into how chemical intermediates contribute to modern industrial and scientific developments.
Chemical Structure of Benzyl Chloride
The structure of benzyl chloride consists of a benzene ring attached to a methylene group that is bonded to a chlorine atom. This arrangement gives the compound unique chemical behavior compared to many other organic substances. The benzene ring provides stability and aromatic characteristics, while the chlorine atom attached to the side chain makes the molecule highly reactive. This combination allows benzyl chloride to participate in various chemical reactions. Its molecular formula is commonly written as C7H7Cl, representing the arrangement of carbon, hydrogen, and chlorine atoms. The structural features of benzyl chloride influence how it interacts with other compounds during chemical reactions.
Physical Properties of Benzyl Chloride
Benzyl chloride is typically found as a colorless to pale yellow liquid with a strong and distinctive odor. The compound has a moderate boiling point and is slightly soluble in water, although it dissolves more easily in many organic solvents. These physical characteristics make benzyl chloride useful in chemical processing environments. Because of its reactive nature, it must be handled carefully in laboratory and industrial settings. The compound’s stability under normal conditions allows it to be stored and transported for industrial use. Understanding the physical properties of benzyl chloride helps chemists manage its safe handling and practical applications.
Production Methods in Chemical Industry
Industrial production of benzyl chloride usually involves the chlorination of toluene. In this process, chlorine gas reacts with toluene under controlled conditions to replace a hydrogen atom in the side chain with a chlorine atom. The reaction is typically carried out using heat or light to promote the chemical transformation. This method allows manufacturers to produce benzyl chloride efficiently on a large scale. The resulting compound is then purified before being used in further chemical reactions. Production techniques are designed to ensure consistency and quality because benzyl chloride often serves as an important intermediate in many industrial products.
Role as a Chemical Intermediate
Benzyl chloride is widely recognized as an essential intermediate in organic chemistry. Its reactive chlorine atom makes it capable of forming new compounds through substitution reactions. Chemists use benzyl chloride to create a variety of products by replacing the chlorine atom with other chemical groups. This versatility allows it to serve as a starting material for many industrial chemicals. Because of this property, benzyl chloride is frequently used in laboratories and manufacturing facilities that produce pharmaceuticals, fragrances, and specialty chemicals. Its role as an intermediate highlights how a single compound can support the creation of many different chemical products.
Applications in Pharmaceutical Manufacturing
One of the most significant uses of benzyl chloride is in the pharmaceutical industry. The compound helps produce various medicinal substances by acting as a building block in chemical synthesis. Researchers and manufacturers rely on benzyl chloride to create compounds that later become active ingredients in certain medications. Its ability to react easily with other molecules makes it valuable in designing complex chemical structures required in drug development. Pharmaceutical companies often use benzyl chloride during early stages of synthesis when forming intermediate compounds. This application demonstrates the compound’s importance in supporting modern medical and healthcare advancements.
Use in Dye and Fragrance Production
Benzyl chloride also plays an important role in the production of dyes and fragrance compounds. In dye manufacturing, it helps create chemical structures that provide color stability and brightness in textile materials. The compound is also used in the preparation of aromatic substances that contribute to fragrances and flavoring agents. Because benzyl chloride contains a benzene ring, it can participate in reactions that produce aromatic derivatives commonly used in these industries. These applications show how benzyl chloride contributes not only to industrial chemistry but also to products used in everyday life such as clothing and scented items.
Safety and Handling Considerations
Due to its reactive nature, benzyl chloride must be handled with proper safety precautions. The compound can cause irritation if it comes into contact with skin, eyes, or respiratory systems. Laboratories and industrial facilities follow strict safety guidelines when working with benzyl chloride to minimize exposure risks. Protective equipment, proper ventilation, and controlled storage conditions are essential when handling the substance. Workers are trained to manage the compound carefully to prevent accidental exposure. Understanding the safety aspects of benzyl chloride ensures that it can be used effectively in chemical processes while maintaining safe working environments.
Environmental Considerations
The production and use of benzyl chloride require attention to environmental responsibility. Chemical manufacturers often implement systems that limit emissions and manage waste generated during production processes. Proper disposal methods are necessary to prevent contamination of soil and water sources. Researchers also explore improved manufacturing techniques that reduce environmental impact while maintaining efficiency. Environmental awareness has become an important part of chemical industry practices. By managing benzyl chloride responsibly, companies can continue benefiting from its industrial value while protecting natural ecosystems and supporting sustainable development goals.
Importance in Organic Chemistry Research
Benzyl chloride holds an important place in organic chemistry research because it provides insight into reaction mechanisms and chemical transformations. Scientists use it in experiments to study substitution reactions and understand how aromatic compounds behave under different conditions. These studies contribute to the development of new chemical methods and materials. Research involving benzyl chloride also helps students and professionals gain practical experience with reactive organic compounds. Its consistent reactivity and well understood structure make it a useful model compound for exploring advanced concepts in chemical science and laboratory experimentation.
Conclusion
Benzyl chloride remains an important compound in modern chemistry due to its versatile properties and wide range of applications. From pharmaceutical manufacturing to dye production and research laboratories, it plays a significant role as a chemical intermediate. Its unique structure, physical characteristics, and reactivity allow scientists and industries to create numerous valuable products. At the same time, responsible handling and environmental awareness are essential to ensure its safe use. By understanding the chemistry and applications of benzyl chloride, readers can appreciate how fundamental chemical compounds contribute to technological progress and industrial innovation across multiple fields.